Hyperliquid Strategies Inc and Sonnet BioTherapeutics Holdings, Inc. Announce Closing of Business Combination
Trading on The Nasdaq Capital Market under the ticker symbol “PURR”
Trading on The Nasdaq Capital Market under the ticker symbol “PURR”
Our modular FHAB construct utilizes a validated, targeted albumin-binding approach to deliver cytokine payloads with enhanced penetration, retention and local activation directly to the tumor.
Sonnet’s deep knowledge of immune biology is complemented by our extensive drug discovery and development expertise.
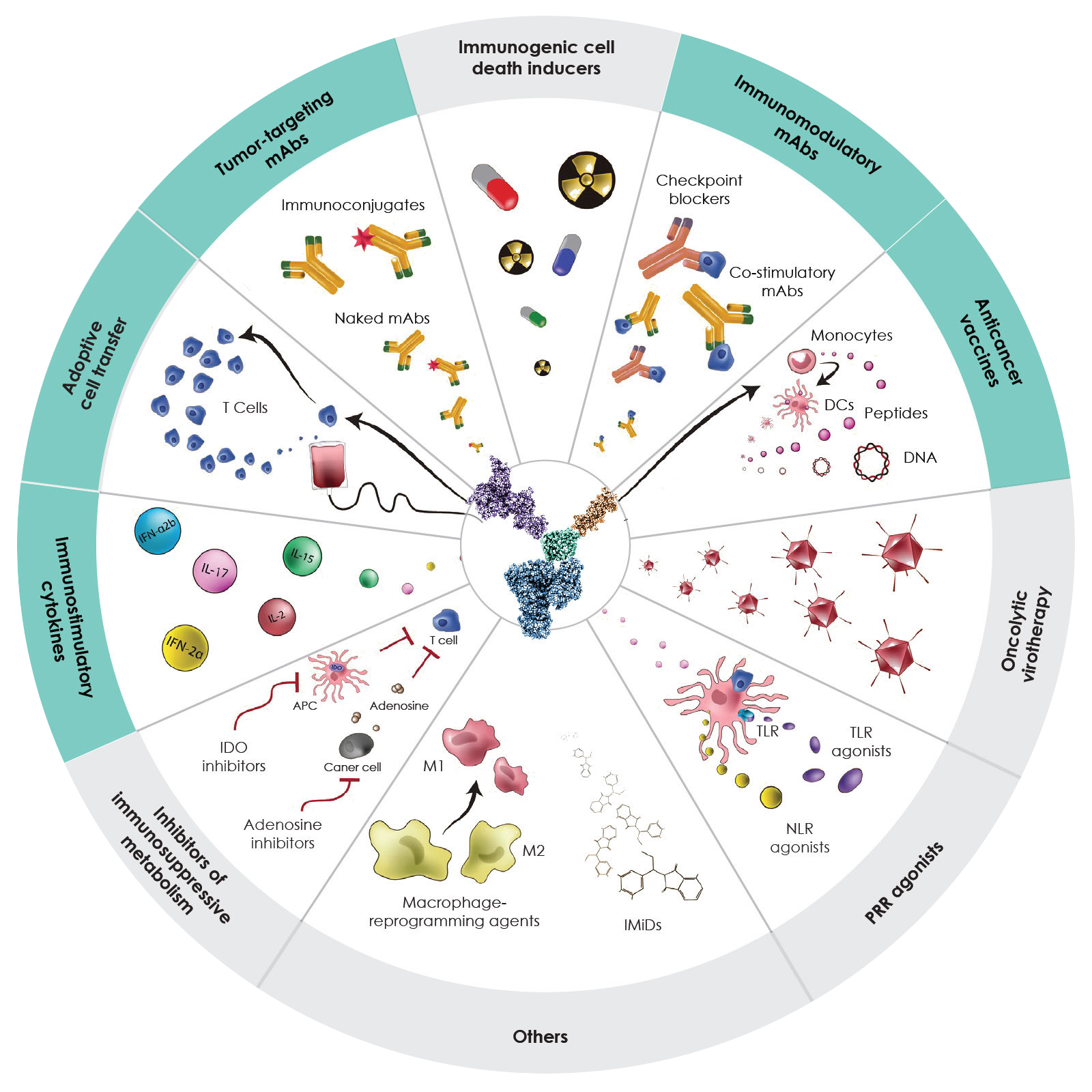
Our Fully Human Albumin-Binding (FHAB) construct is the foundation of a fully modular technology that enables the design of single or bifunctional biologic compounds that target albumin, which binds to FcRn, GP60 and SPARC.
Human Serum Albumin (HSA) is naturally present in the bloodstream and is the predominant protein in blood plasma. Albumin accumulates at sites of inflammation, including tumors. Following administration, Sonnet’s FHAB-conjugated therapies bind to and “hitch-hike” on patients’ endogenous HSA for transport to the target tissues.
